- europages
- >
- COMPANIES - SUPPLIERS - SERVICE PROVIDERS
- >
- price negotiations
Results for
Price negotiations - Import export
EC PLAZA
South Korea
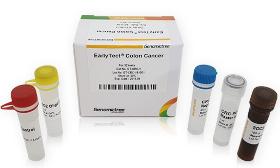
Product Description EarlyTect Colon Cancer Test was approved by the Korea Ministry of Food and Drug Safety through clinical trials at Yonsei Medical Center, Severance Hospital, and Severance Check-up Center for eligible subjects, 585 men and women aged 30 to 80. Clinical trials using fecal DNA demonstrated that CRC can be diagnosed with 90% sensitivity and specificity. EarlyTect Colon Cancer Test is a qualitative real-time PCR test that measures methylated Syndecan-2, a biomarker clinically related to precancerous lesions of CRC, in stool DNA to diagnose colorectal cancer. This product is not intended to confirm colorectal cancer, so if a subject was positive, CRC or precancerous lesions of CRC may exist. Product Standard - Cancer Type: Colorectal Cancer - Subject: Subjects for asymptomatic colorectal cancer screening - Biomarker: SDC2 me The price and transaction conditions are negotiable. Please Request a Quote for the transaction
Request for a quote
EC PLAZA
South Korea
<Product Features> Precision processing using various materials Ceramic quartz sapphire zirconia etc. <Product Specifications> We process and supply a variety of materials according to customer needs. <Product Use> machine chemistry optics <Remark> Purchase Price and Terms are negotiable. <Company Information> GEO Co., Ltd Founded as GEO Trading in 1996, we continuously developed and ungraded ourselves to become GEO Corporation. Based on the experience of importing and exporting Fine ceramics and optical products, we are now dealing with sapphire, semiconductor, and optical quartz in domestic market. GEO Corp. will endlessly strive to pioneer the overseas markets of fine ceramics and automatic door sensors. Website : www.geocorp.co.kr Tel : +82 32-323-6650
Request for a quoteDo you sell or make similar products?
Sign up to europages and have your products listed
Results for
Price negotiations - Import exportNumber of results
2 ProductsCountries
Company type
Category