AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Manufacturer/ Producer


AMETEK ENGINEERED MEDICAL COMPONENTS
United States
The ablation of fine wires by laser technology is a precise technique that involves the controlled removal of material from conductive wires, enabling numerous applications across various industries.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Nitinol laser processing, involving the use of lasers for cutting, welding, or shaping Nitinol alloy, offers several advantages, especially in the medical device industry.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Laserage is widely regarded as the medical industry-leading expert in laser contract manufacturing. Since 1979, we have been leading the way in the field of custom laser processing, providing medical device component fabrication with numerous laser manufacturing capabilities.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Stent manufacturing applies to a wide variety of medical applications including cardiovascular applications, birth control, kidney stone pain control and esophageal and gastrointestinal uses.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
AMETEK EMC offers technical expertise and precision engineering to support customers in the development and manufacture of the most demanding applications.
Request for a quote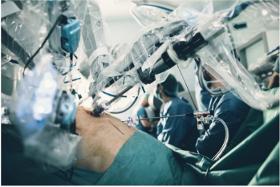
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Robotic surgery is considered a minimally invasive surgery carried out using mechanical arms that have surgical instruments as well as a camera arm attached which are controlled by the surgeon seated at a computer console near the operating table.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Cardiac rhythm management (CRM) means artificially controlling and monitoring the heartbeat. This is usually done through small battery-powered devices that are implanted into the human body and connected to the heart. The two most common devices that manage the heart rhythm are pacemakers and cardioverter defibrillators (ICD).
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
We use Silicone Molding at the end of cable assemblies to connect the cable with the connectors. Along with manufacturing over-molding alternatives for limited or single-use devices, we use our over-molding for the components that humans come in contact with to improve the feel and touch of the device, such as molding and overmolding for ECG cables.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
There are multiple advantages that LSR offers over plastic injection molding to the medical device industry and in patient care.
Request for a quote
AMETEK ENGINEERED MEDICAL COMPONENTS
United States
Silicone transfer molding is another manufacturing process used in the medical device industry. It is similar to LSR molding but focuses on more delicate electrical connections or instruments. It is a 2-step process that involves the manual molding of components into shape.
Request for a quoteManufacturer/ Producer
7800 Equitable Drive
Ste 250
55344 Eden Prairie - United States
europages also recommends
A selection of companies related to the activity:
A selection of products that might interest you

ANSMANN AG
Germany
The new IPC charger series is the latest innovation in the field of quick-charging. The universal chargers have integrated communication and support all popular communication protocols, such as CAN, I2C, SM, LIN, HDQ. Li-ion rechargeable battery packs from 24 V to 48 V will be automatically detected. Once the E-BUS charger has made contact with the rechargeable battery, an extensive diagnostics procedure is started. The intelligent chargers identify the rechargeable battery voltage, the charge current is adapted and the charge process started. The output power here is up to 300 W. This enables the shortest possible charge times with simultaneous exchange of parameters to be realised with applications such as in drive systems, power tools or medical equipment. With the IPC 300W we are delighted to present a universal charger that can adapt to all lithium ion rechargeable battery systems.
Request for a quote
ANSMANN AG
Germany
Planning a new development or a redesign of a rechargeable battery or charger? ANSMANN – Your partner from the initial idea through the planning, design, development and manufacturing stages and on to serial manufacturing. Based on your application and the requirements, we can guide you with professional advice for the selection of the correct cells, the capabilities of the necessary protective circuitry, compliance with standards and certification (as early as in the development phase), the first prototypes as STL parts and the first measurements. We can support you through all of the important steps towards your final decision. We can take on the design and manufacturing of the housing parts and the injection and stamping tools required for this. Single-source delivery and manufacturing guarantees optimum interaction between rechargeable battery, BMS and charger. The cells that we use are delivered by renowned manufacturers such as Panasonic, LG, Samsung, Sanyo, A123, Sony, etc.
Request for a quote
SOL-MILLENNIUM
Poland
Non - sterile single use polypropylene holder for safe and effective blood collection to blood culture bottles.
Request for a quote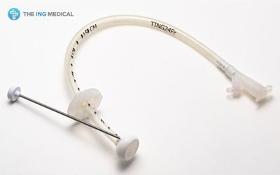
EC PLAZA
South Korea
1) Product Description The Gastrostomy Feeding Tube are indicated for use in patients who require long term feeding, are unable to tolerate oral feeding, who are at low risk for aspration, require gastric decomparession. It is a tube used to supply food and drugs to the stomach. 2) Product Features - Easy and less traumatic to insert due to the soft and compressible internal dome. - Radiopaque dome allows x-ray placement confirmation - Identification of tube obstructions enhanced with translucent tubings - Soft, less traumatic silicone designed for patient comfort
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free



