- europages
- >
- Medical Equipment
- >
- SCREEN ITALIA
- >
- Screen Check Test Covid-19 Antigen
Screen Check Test Covid-19 Antigen
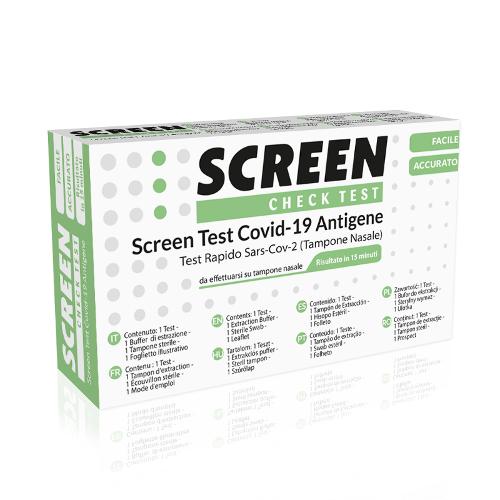
Description
The rapid test for self-diagnostic use for SARS-CoV-2 antigen is a single-use in vitro immunochromatographic test designed for rapid qualitative detection of SARS-CoV-2 virus antigens. Screen Test Covid-19 Antigen is an in vitro diagnostic medical device CE 1434. Read the warnings and operating instructions carefully.
- Medical Equipment
- Covid Test
- Screen Test
- Antigen Test
Similar products from SCREEN ITALIA
SCREEN ITALIA
Italy
Antigen Combo Rapid Test – Product code: ISIR-535(SC-2399-20) SARS-CoV-2/Influenza A+B/RSV Antigen Combo Rapid Test is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2 Nucleocapsid protein, Influenza A, Influenza B and Respiratory Syncytial Virus(RSV) antigens present in human nasopharynx. For professional in vitro diagnostic use only. The SARS-CoV-2/Influenza A+B/RSV Antigen Combo Rapid Test (Nasopharyngeal Swab) is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2 Nucleocapsid protein, Influenza A, Influenza B and Respiratory Syncytial Virus(RSV) antigens in nasopharyngeal swab specimens from individuals with suspected SARS-CoV-2/Influenza/RSV infection in conjunction with clinical presentation and the results of other laboratory tests. Results are for the detection of SARS-CoV-2 Nucleocapsid protein, Influenza A+B and RSV Antigens.
Request for a quote
SCREEN ITALIA
Italy
Single-use self test designed for the detection of the novel coronavirus Sars-CoV-2 in human oral fluid. The Rapid COVID-19 Antigen Test (Oral Fluid) is a membrane-based qualitative immunoassay for the determination of SARS-CoV-2 antigens in human oral fluid samples. Result in 15 minutes. Screen Check Test Covid-19 Saliva is an in vitro diagnostic medical device CE 1434. Read the warnings and instructions carefully.
Request for a quote
SCREEN ITALIA
Italy
The rapid Screen Test Igg Sars-Cov-2 S-RBD test from whole prick blood is a rapid immunochromatographic test for the qualitative detection of IgG antibodies to the receptor binding domain (RBD) spike (S) protein of SARS-CoV-2 in human whole prick blood at approximately 10 days after vaccination. Screen Test Covid-19 Igg Sars-Cov-2 S-RBD is an in vitro diagnostic medical device CE 1434. Read the warnings and operating instructions carefully.
Request for a quote
SCREEN ITALIA
Italy
Mycoplasma Pneumoniae Antigen Test – Product code: SC-0654-25 Rapid test for the qualitative detection of Mycoplasma Pneumoniae Antigen on a pharyngeal sample. For professional in vitro diagnostic use only. The rapid test for Mycoplasma Pneumoniae Antigen is a rapid chromatographic immunoassay for the qualitative detection of mycoplasma pneumoniae (M. Pneumoniae) antigens in human throat swabs. It is intended to assist in the rapid differential diagnosis of mycoplasma pneumoniae infections. M. pneumoniae is one of three Mycoplasma species that frequently cause infections in humans. M. pneumoniae most commonly causes upper respiratory tract infections, but it can also cause pneumonia. The identification of M.pneumoniae will help in the treatment of the disease with appropriate antibiotic therapy. This immunoassay for M. pneumoniae is designed to qualitatively detect the M. pneumoniae antigen.
Request for a quote