- europages
- >
- Medical Equipment
- >
- AKIN GLOBAL MEDICAL
- >
- Hartmann Hydrocoll
Hartmann Hydrocoll

Description
Hydrocoll, Permafoam, Atrauman all codes available. Brand-new.
- Medical Equipment
- hartmann
- hydrocoll
- atrauman
Similar products

GENJECT
Turkey
Genject brand disposable needles are made of stainless steel. It is produced in accordance with TS EN ISO 7864 – TS EN ISO 9626 standards. All of our needles are sterile and non-toxic. Our products have transparent plastic protection.
Request for a quote
SAM3D
France
Marking key points on the body - Photographs of different positions - A personalised assessment is given to each patient
Request for a quote
GERWING MEDIZINPRODUKTE GERD RIESTER E.K.
Germany
Tracheo-FIX, cannula retaining strap, for tracheostomies, in two parts, with elastic element 2,5cm wide
Request for a quote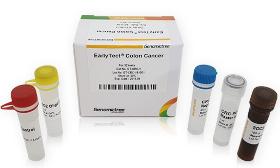
EC PLAZA
South Korea
Product Description EarlyTect Colon Cancer Test was approved by the Korea Ministry of Food and Drug Safety through clinical trials at Yonsei Medical Center, Severance Hospital, and Severance Check-up Center for eligible subjects, 585 men and women aged 30 to 80. Clinical trials using fecal DNA demonstrated that CRC can be diagnosed with 90% sensitivity and specificity. EarlyTect Colon Cancer Test is a qualitative real-time PCR test that measures methylated Syndecan-2, a biomarker clinically related to precancerous lesions of CRC, in stool DNA to diagnose colorectal cancer. This product is not intended to confirm colorectal cancer, so if a subject was positive, CRC or precancerous lesions of CRC may exist. Product Standard - Cancer Type: Colorectal Cancer - Subject: Subjects for asymptomatic colorectal cancer screening - Biomarker: SDC2 me The price and transaction conditions are negotiable. Please Request a Quote for the transaction
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free