- europages
- >
- Medical tests
- >
- ASCEND CARE LTD
- >
- £2: Humasis COVID Antigen Lateral Flow Test: HCP X25
£2: Humasis COVID Antigen Lateral Flow Test: HCP X25
Health Care Professional Only
Description
The Humasis COVID-19 Test is an in vitro diagnostic test based on lateral flow chromatographic immunoassay intended for qualitative detection of SARS-CoV-2 antigen in human nasopharynx. For healthcare professional use in near patient testing environments. Sensitivity 95.5% & Specificity 100% Kit Contents ▪ 25 x Test devices packaged individually in aluminium pouch ▪ 25 x Disposable test tubes with extraction buffer ▪ 25 x Filter caps ▪ 25 x Sterilised swabs for specimen collection ▪ 1 x Instructions for use *£2 ex vat per test *All prices are ex vat and shipping costs Certificates: FDA European Commission CE Marked Porton Down DHSC Manufacturer Humasis Co., LTD, South Korea EN ISO 13485:2016 Detection COVID-19 Antigen Method Immunochromatographic Assay Test Type Cassette Package 25T/Kit Specimen Nasopharyngeal or Nasal Swab Shelf Life 18 Months Storage 2-30 Celsius Test Time 15 minutes Cat No. ACOVA-7025
- Medical tests
- IVD Diagnostic COVID LFT's
- covid 19
- Lateral Flow Tests
Similar products

MANNEQUINS SHOPPING
France
Pack of 10 plexiglass protection screens with metal base. Anti-covid protective screen for offices and restaurants Protective screens sold in packs of 10 pieces. Protect your teams and your customers with these plexiglass protective screens.
Request for a quote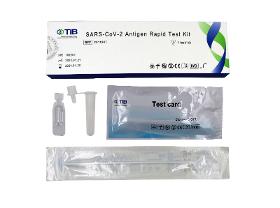
TRIPLEX INTERNATIONAL BIOSCIENCES
China
The SARSCoV2 Antigen Rapid Test Kit is a colloidal gold enhanced double antibody san dwich immunoassay for the qualitative determination of SARSCoV2 antigen.The test region (T) of nitrocellulose membrane was immobilized with mouse anti SARSCoV2 antibody 1 (Ab1), and the control region (C) was immobilized with Goat anti chicken IgY antibody (GAC). At the same time, the binding pad of the test card was fixed with mouse anti SARSCoV2 antibody 2 (Ab2) a nd chicken IgY (CIgY) which were both labeled by colloidal gold.
Request for a quote
EC PLAZA
South Korea
Our company has been growing for 20 years with the goal of One Health. - Mission: Contribute to human and animal health with diagnostic business. - Vision: Company supplying the world’s highest quality diagnostic kits.
Request for a quote
S.A.M MED BV
Netherlands
Syringe Testing is a critical step in developing and commercializing needlebased injection systems for medical use. By testing mechanical properties, such as breakloose and glide force, as well as biocompatibility, extractables and leachables, sterility, packaging and transit properties, we can determine if the device is within standard specifications, reducing the likelihood of improper function. Eurofins Medical Device Testing network of laboratories has experience performing an array of standard and nonstandard testing on syringes to verify their safety. Choose Eurofins Medical Device Testing to help you Determine the necessary testing requirements for your device Write GMPcompliant and ISO standard driven protocols and methods Perform the required tests to demonstrate that the expected performance criteria are satisfied Complete batch release activities and/or gain regulatory approval
Request for a quoteRequest for quotes
Create one request and get multiple quotes form verified suppliers.
- Only relevant suppliers
- Data privacy compliant
- 100% free